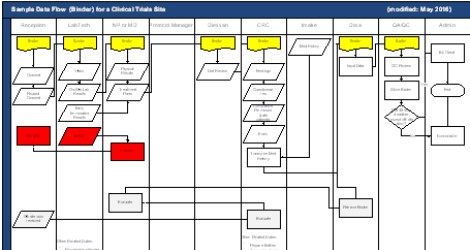
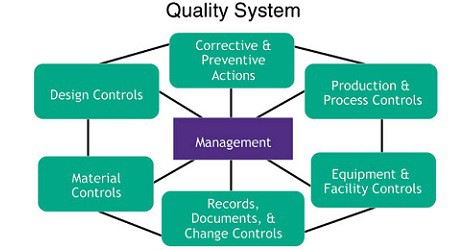
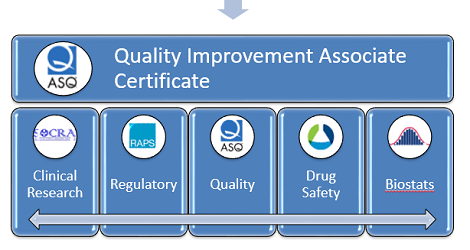
Writing a Resume for Pharma or Biotech Career -Advice from Professional Writer
Writing a Resume for Pharma No matter how technically astute you are, if you can’t sell your strengths during your job search, your job search can run much longer than you like. Here are some tips from Fatemah Mirza, professional resume writer at CareerTuners on writing a resume for pharma or biotech resume: 1) Use […]
read more